
Determination Of Order Of Reaction Study Material For Iit Jee Askiitians
T 1/2 of first order reaction is 10 min Starting with 10 molL −1, rate after min is A molL −1min −1 B ×25 molL −1min −1 C ×5 molL −1min −1 D ×10 molL −1min −1The half life of a first order reaction A → B, is given as (a) t1/2 = 0693 K (b) t1/2 = 0693 In K (c) t1/2 = 0693/K (d) t1/2 log2/K askedin Chemistryby rav(669kpoints) class12
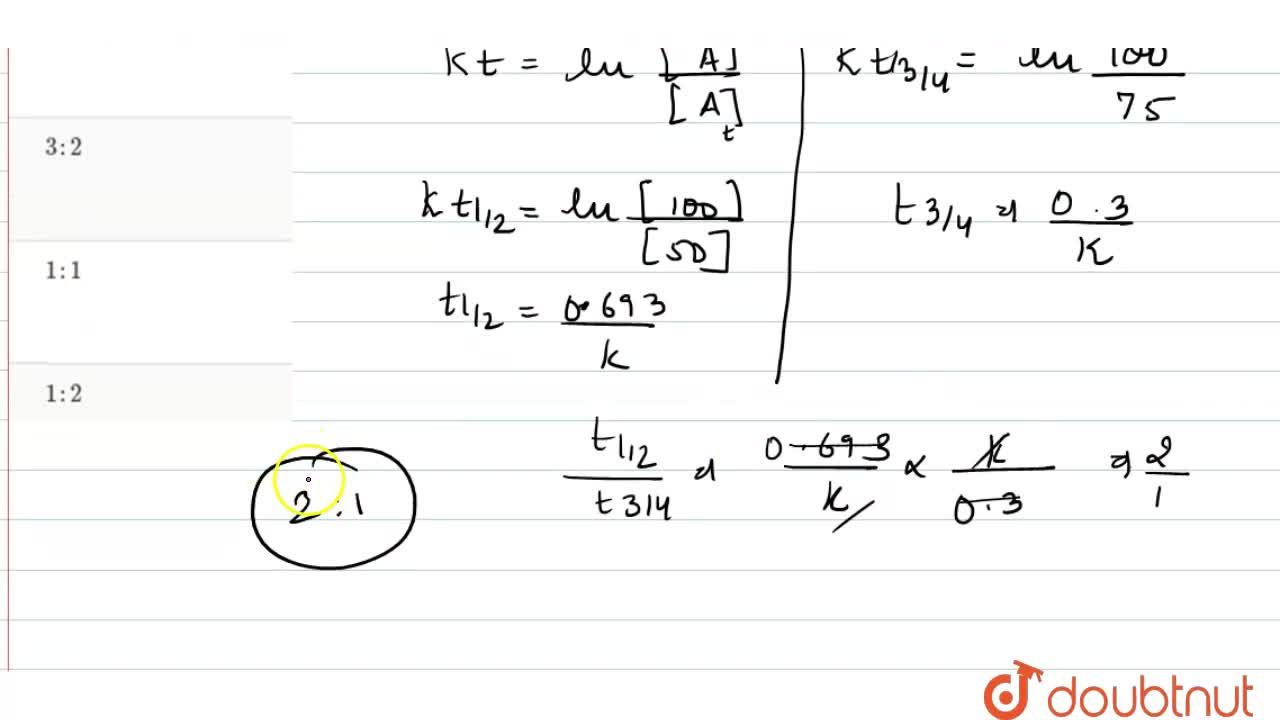
T1/2 for first order reaction is
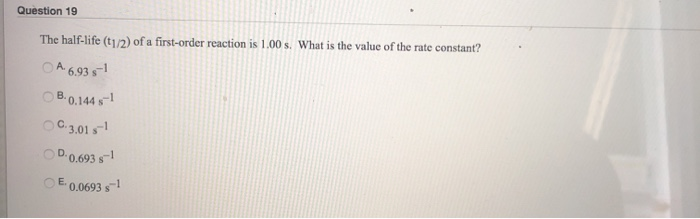
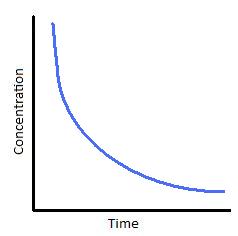
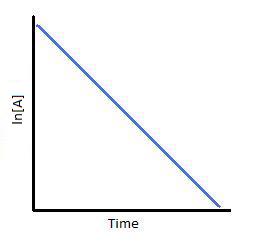
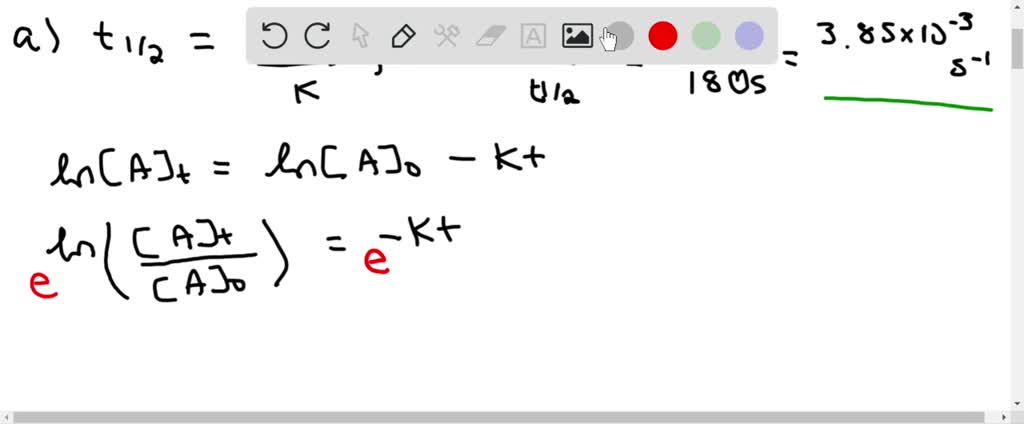
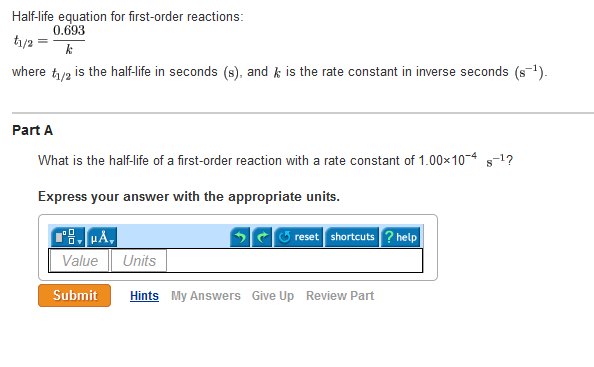
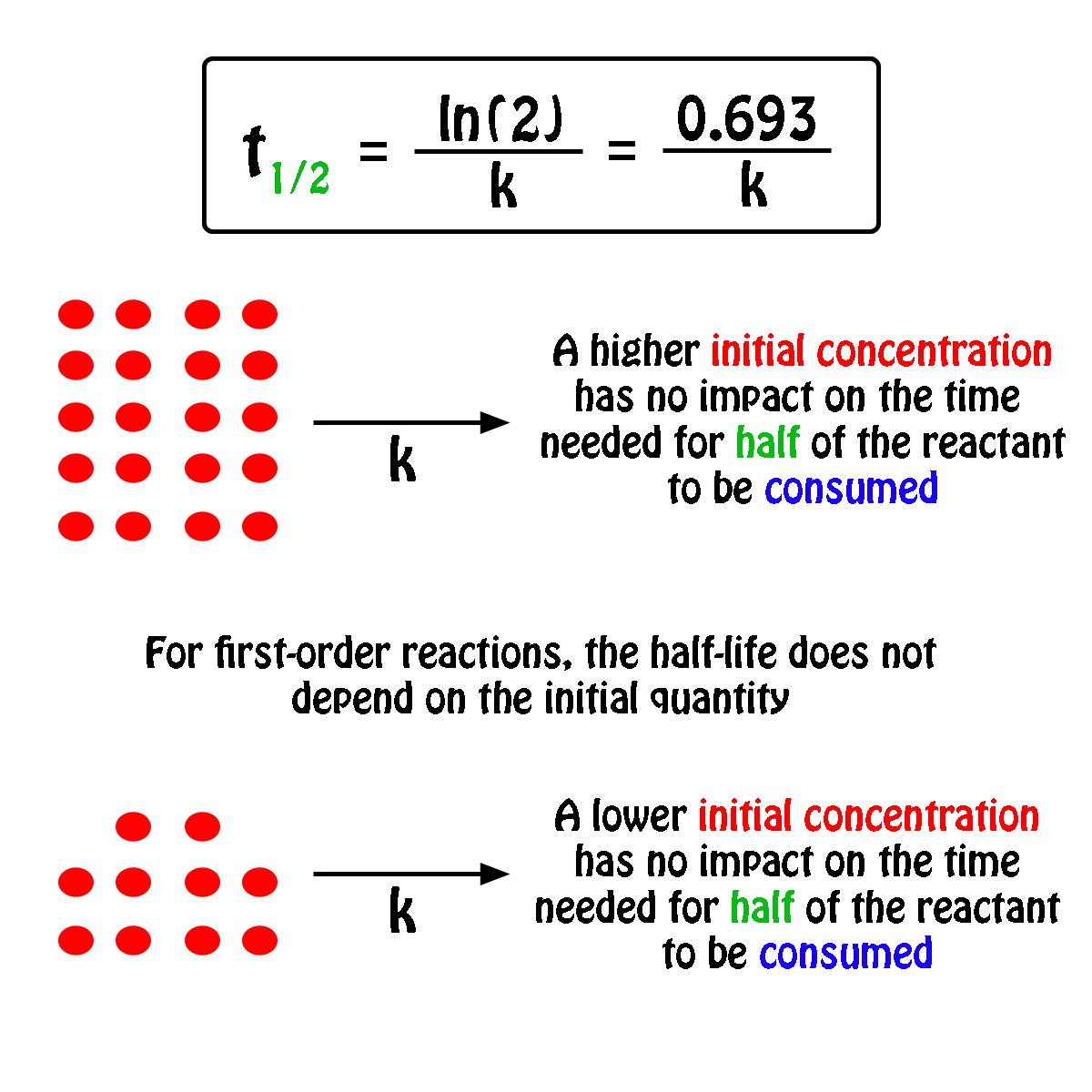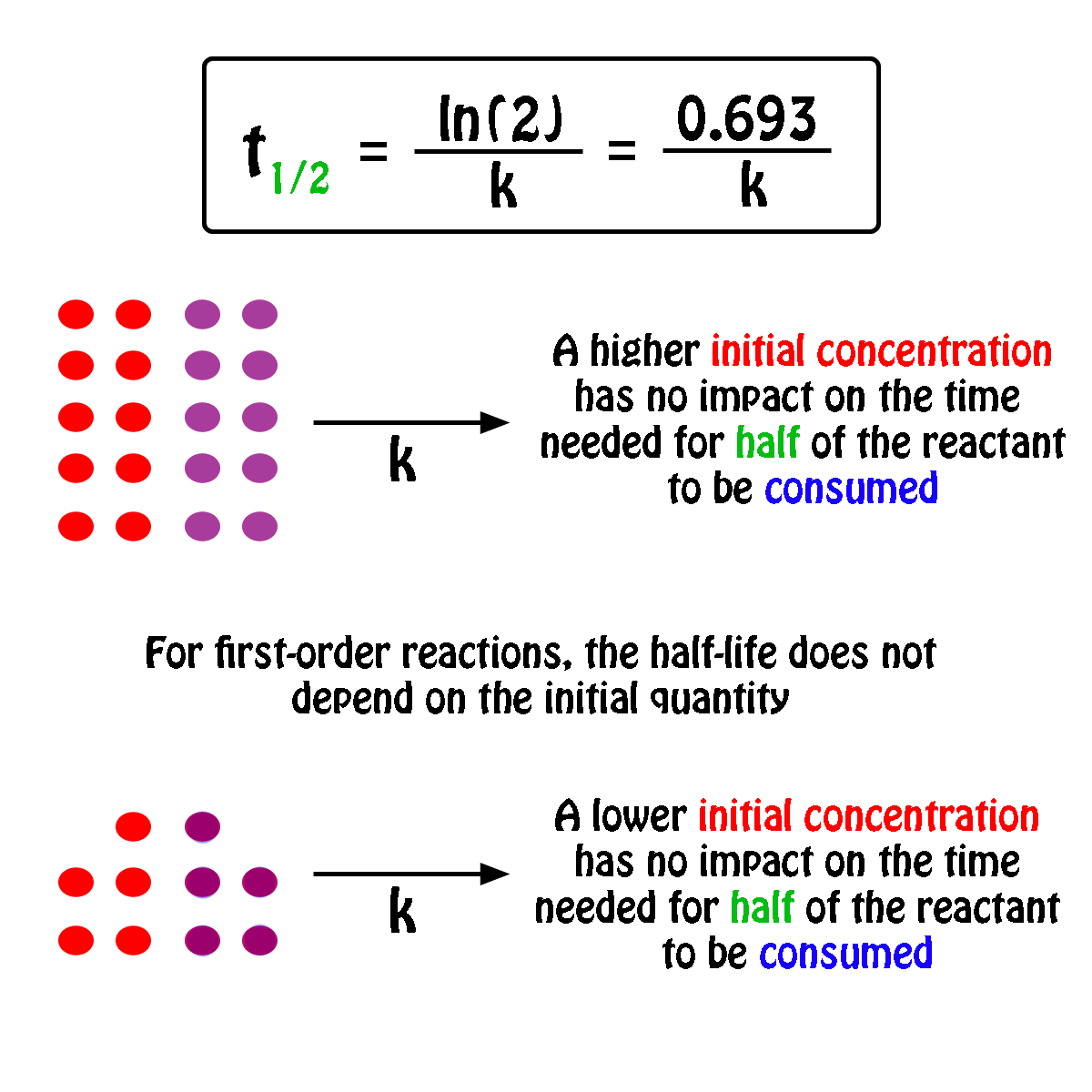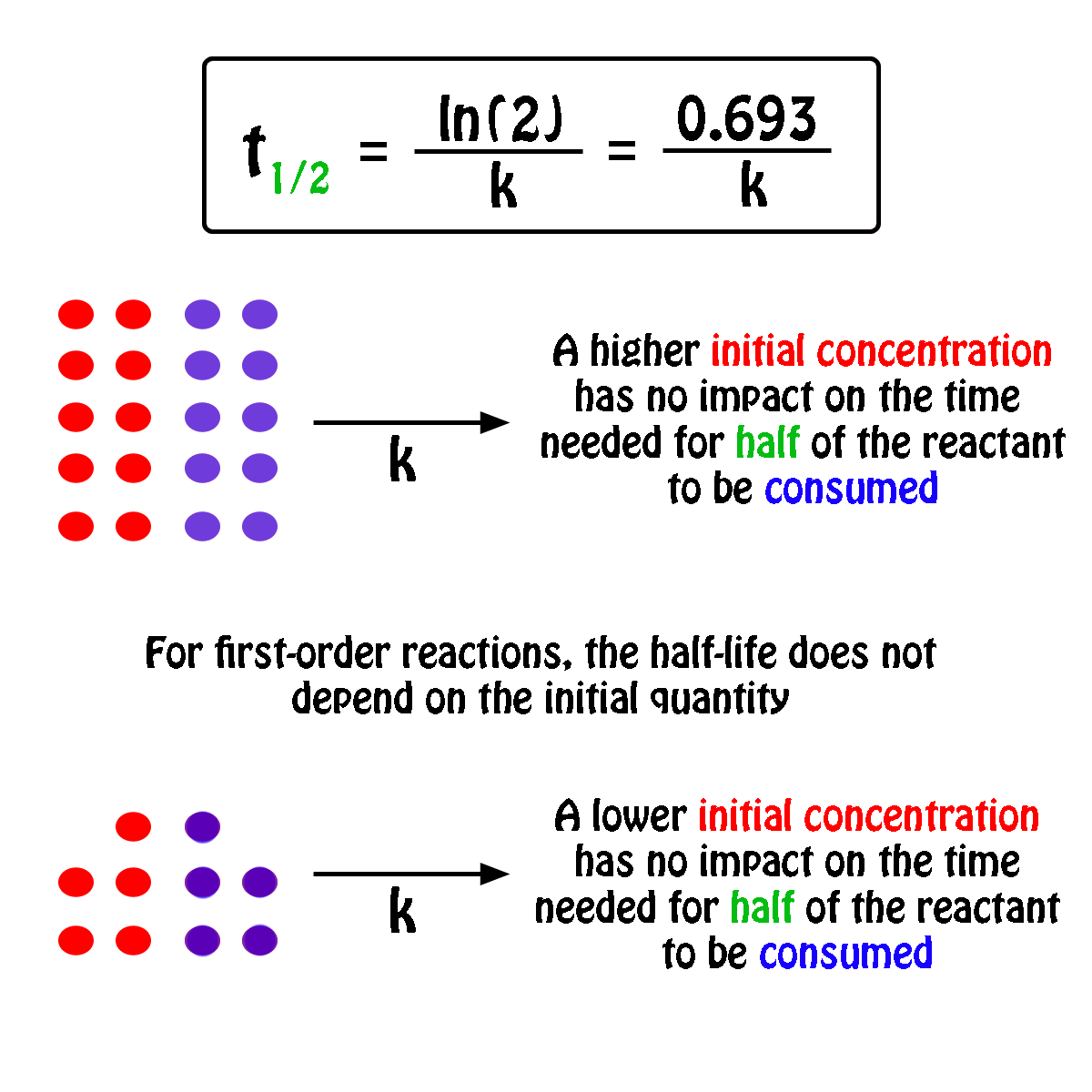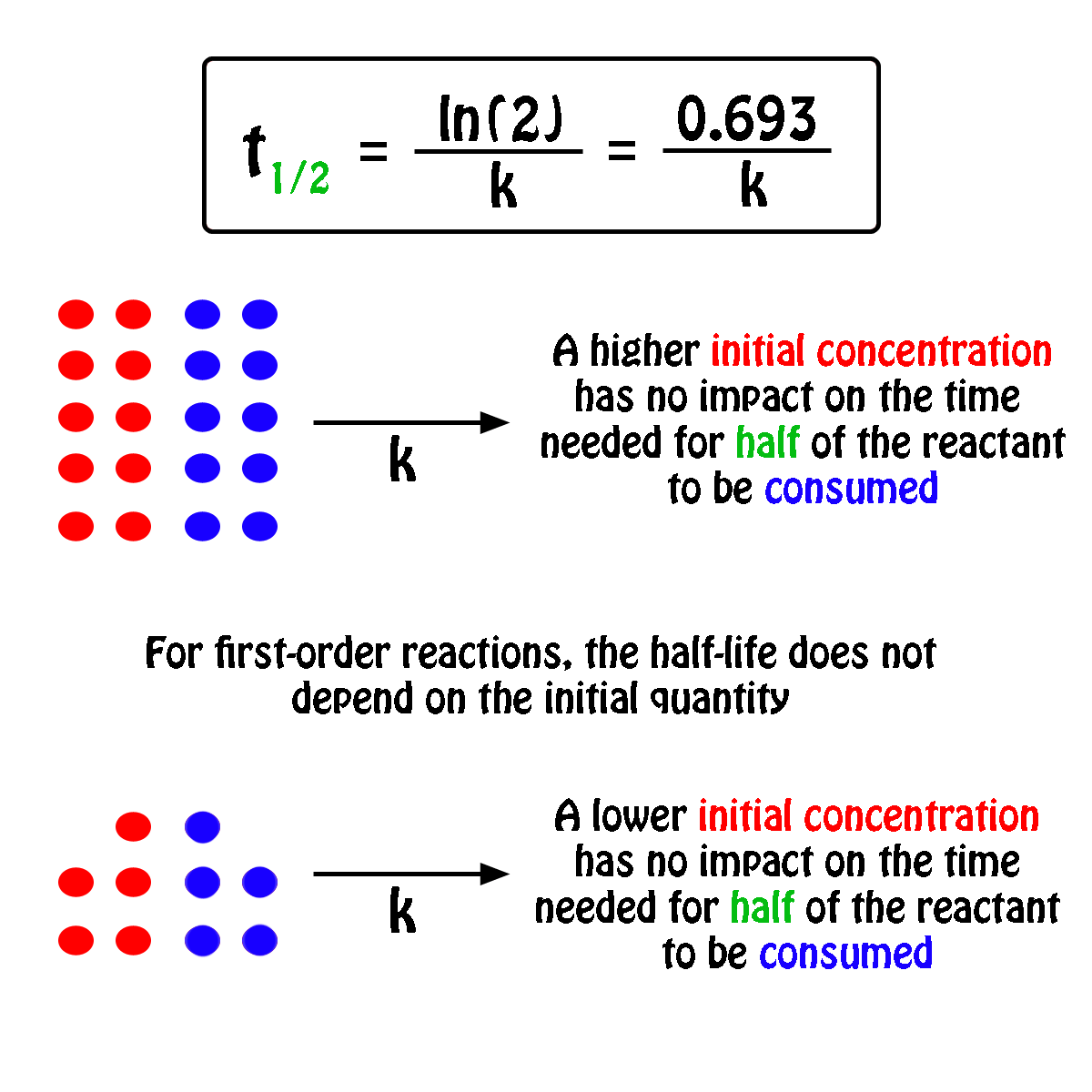
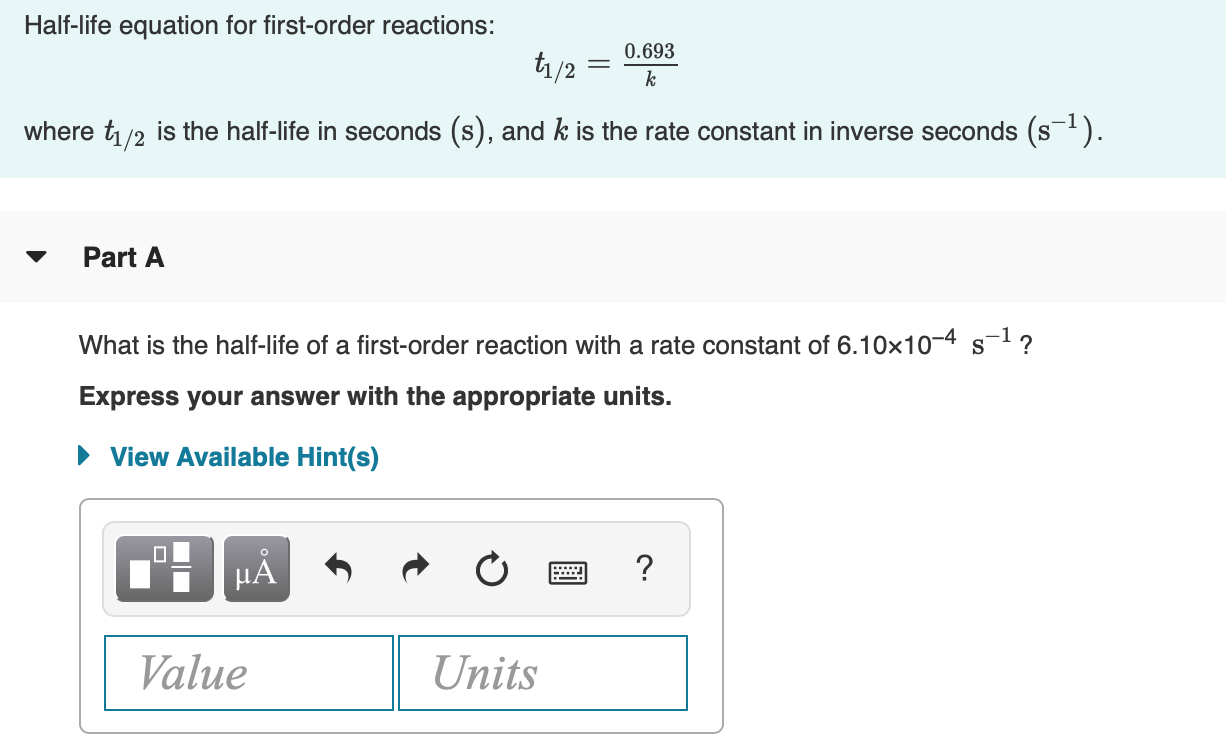
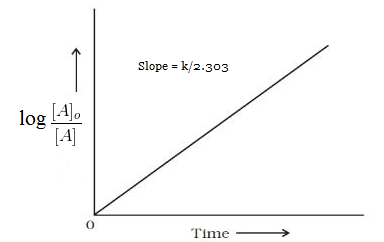
T1/2 for first order reaction is-See the answer Halflife equation for firstorder reactions t1/2=0693k where t1/2 is the halflife in seconds (s), and k is the rate constant in inverse seconds (s−1)For a first order reaction, t 1/2 is thrice the t 7/8 For a first order reaction, t 1/2 is independent of the initial concentration of reactants A Both Assertion and Reason are correct and Reason is the
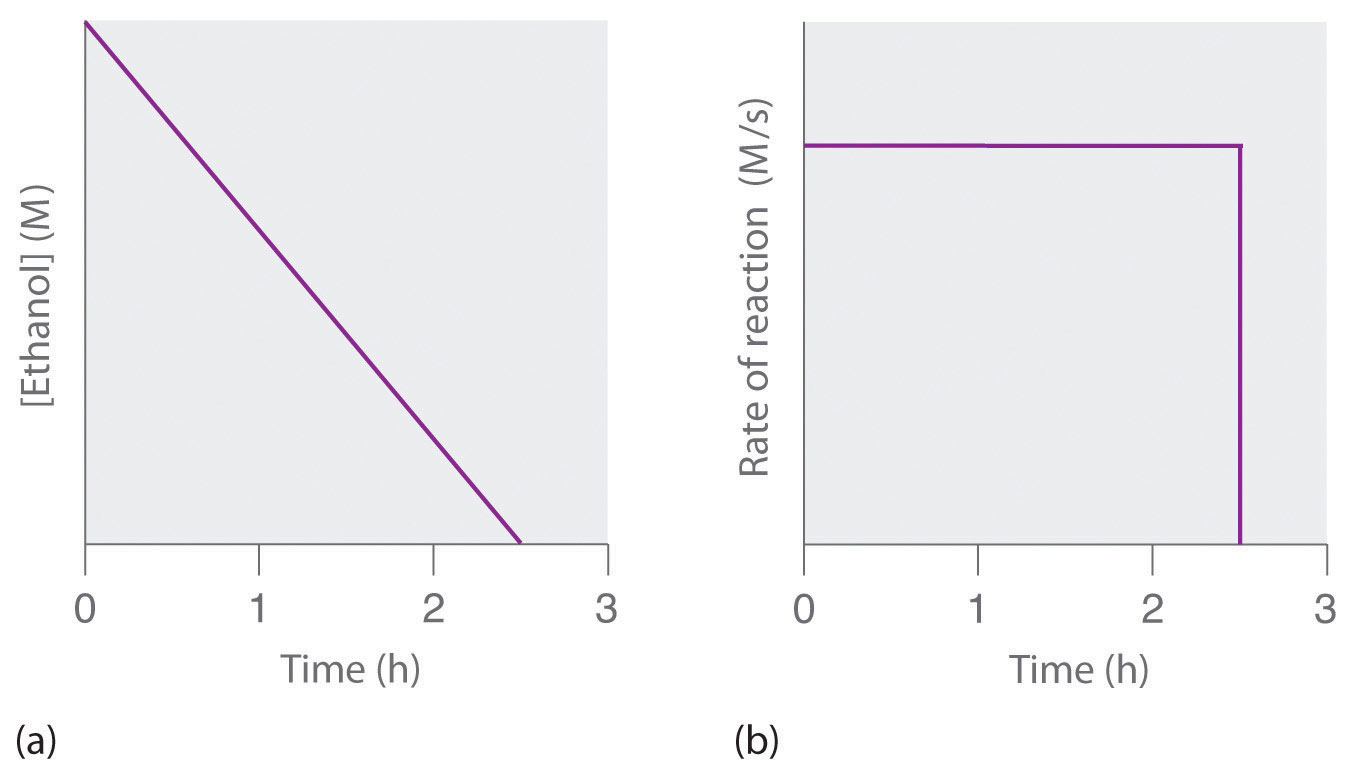
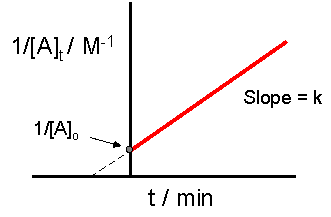
Concentration Time Relationships Integrated Rate Laws Introductory Chemistry
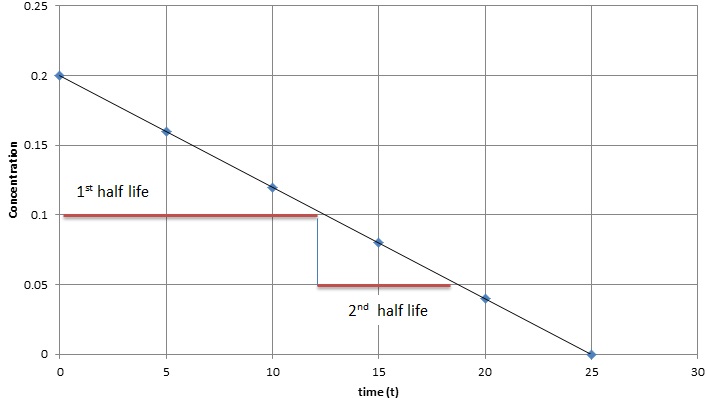
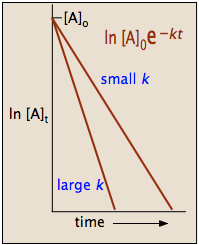
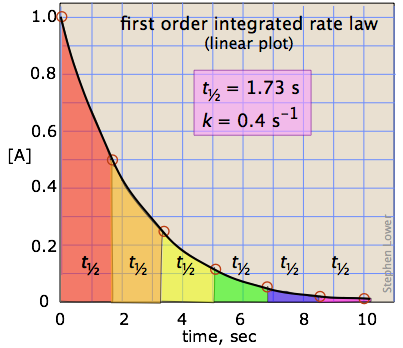
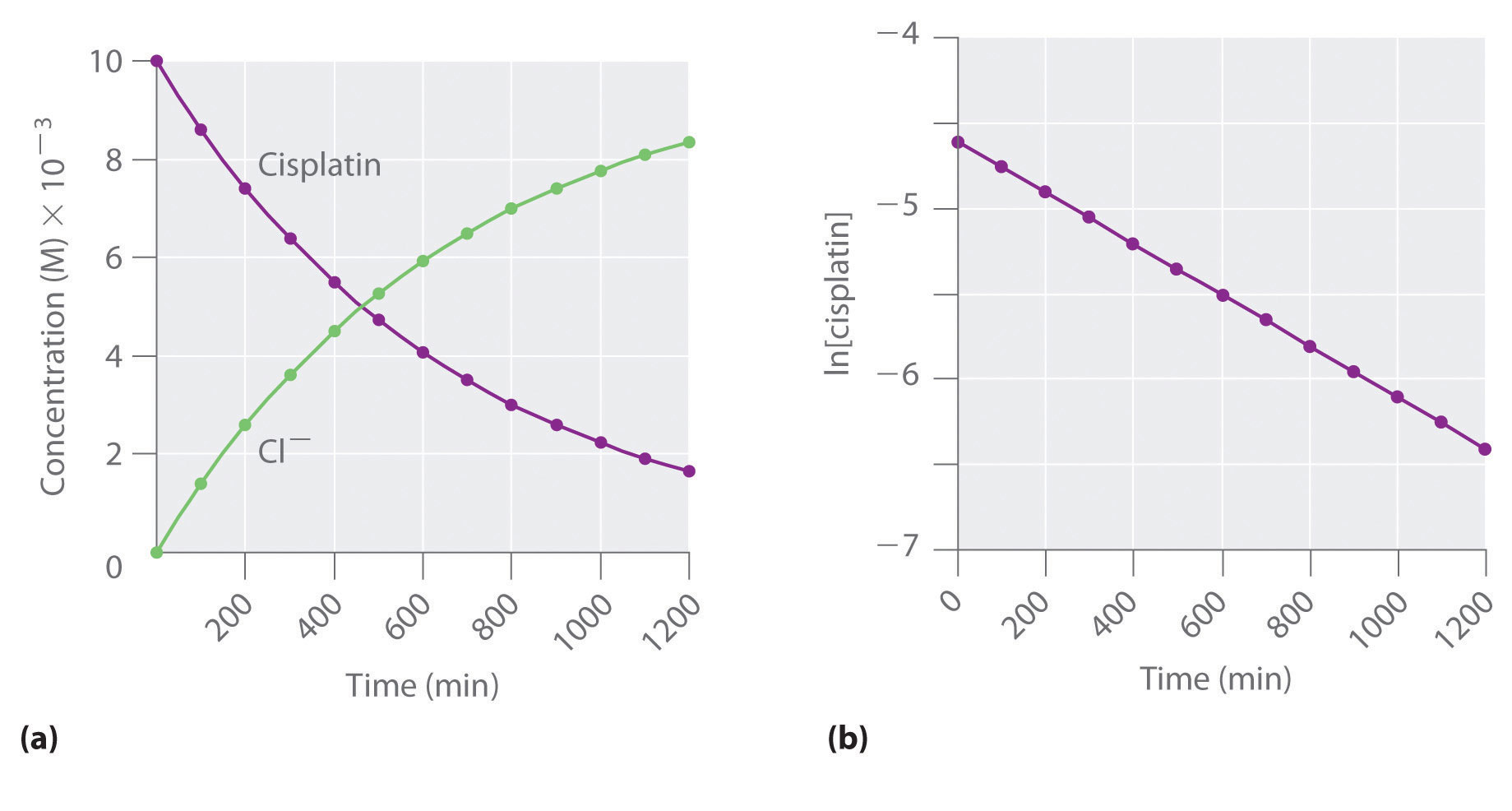
Halflives of first order reactions The halflife ( t1 / 2) is a timescale on which the initial population is decreased by half of its original value, represented by the following equationCalculate t1/2 Solution For a first order reaction, Therefore, t1/2 of the decomposition reaction is = 777 min (approximately) Suggest Corrections 0 Same exercise questions Q Write the NernstAdd to Chrome Home Class 12 Chemistry Chemical Kinetics Chemical Kinetics Question 5 views The t1/2 of first order reaction is 60 minutes
Given, t 1/2 = 10 min t = min N 0 = 10 M ⇒ t = n t 1/2 = n × 10 ∴ n = 2 ⇒ N 0 N = (2 1 ) n ⇒ 10 N = (2 1 ) 2 or 10 N = 4 1 ∴ N = 4 10 = 25 M ∴ Rate = k A = × 25 M min − 1The halflife period of a first order reaction is independent of the initial concentration of the reactant t1/2 =k0693 NEET 22 Chemistry Questions The pHof the solution containing Ans Halflife of a firstorder reaction doesn't rely on the concentration of the reactant It is a constant and is related to the rate constant for the reaction t1/2 = 0693/k
T1/2 for first order reaction isのギャラリー
各画像をクリックすると、ダウンロードまたは拡大表示できます
 | ||
 |  | |
 |  | |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 | ||
 |  |  |
 |  | |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 |  |  |
 |  |  |
 |  |  |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 | 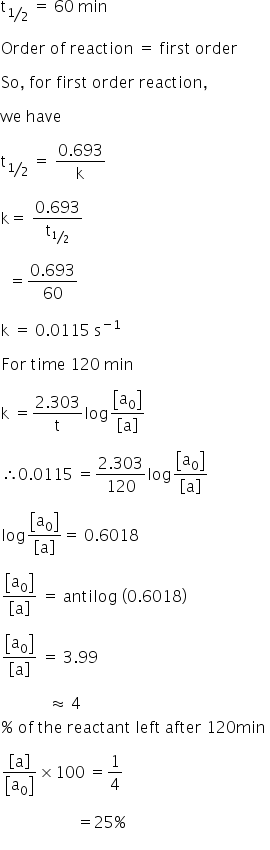 |  |
 |  |  |
 |  |  |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 |  |  |
 |  |  |
 |  | |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 |  |  |
 |  |  |
 |  | |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 |  |  |
 | ||
 |  |  |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
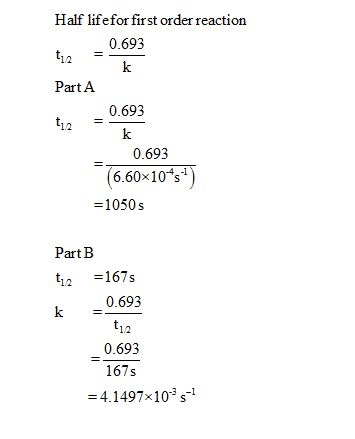 |  | |
 |  |  |
 |  |  |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 | ||
 |  | |
 |  |  |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 |  |  |
 |  | |
 |  | |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 |  |  |
 |  |  |
 |  |  |
「T1/2 for first order reaction is」の画像ギャラリー、詳細は各画像をクリックしてください。
 | ||
 |
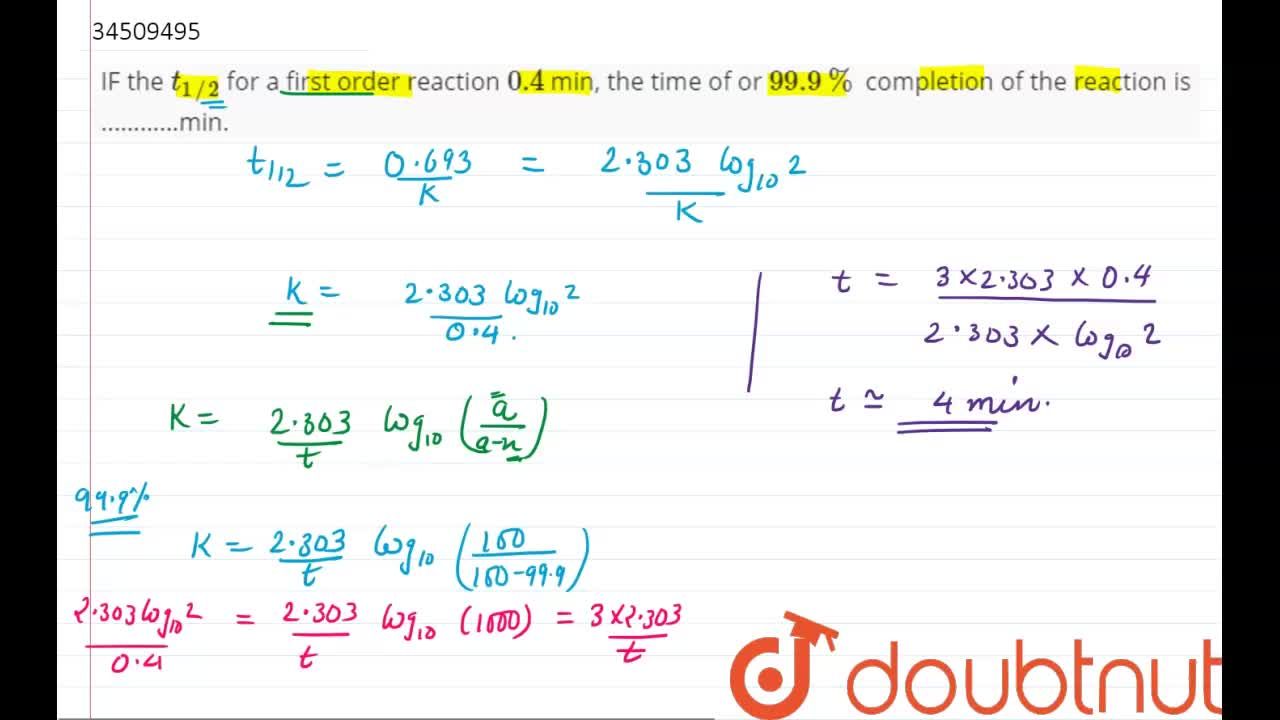
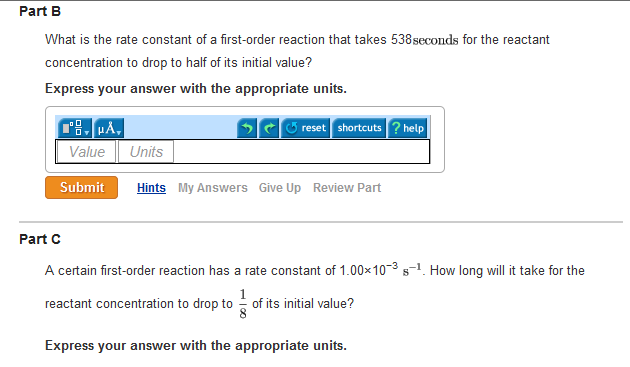
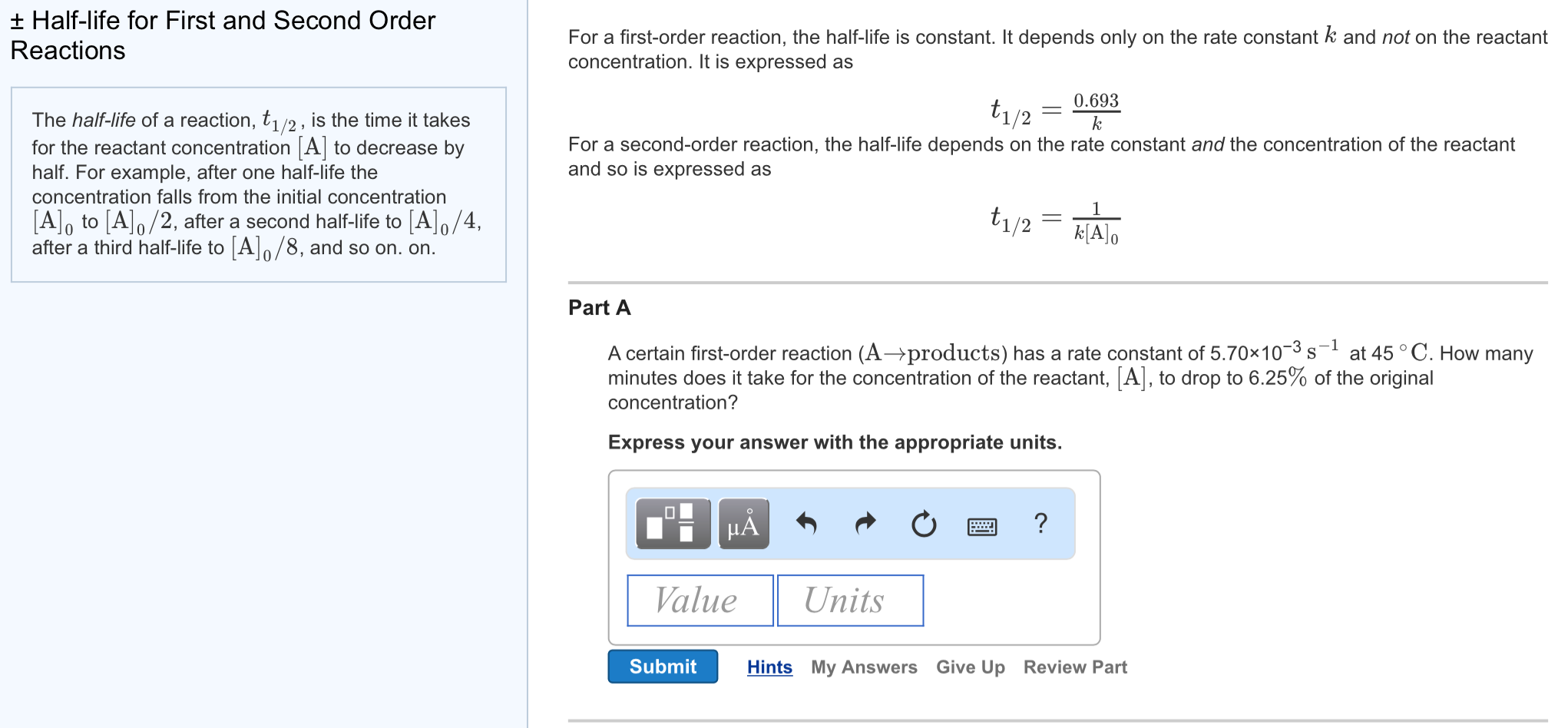
In a first order reaction, time required for completion of 999% is X times of halflife (t1/2) of the reactionWhen reaction is completed 999%, Rn= R0– 0999R0 The value of X is 1 5 2 10Given, First order reaction t1/2 = 0693 K (A) Given rate constant (K) = 870 x 104 s1 To find halflife Solution by using formula t1/2 = 0693/ View the full answer Previous question Next
Incoming Term: t1/2 for first order reaction is 10 minutes, t1/2 for first order reaction is, t1/2 for first order reaction is 14.26 minutes, t1/2 for a first order reaction is 32 minutes calculate t3/4, t1/2 of first order reaction is 60 minutes, the value of t1/2 for first order reaction is, half life period t1/2) for first order reaction is, if t1/2 of first order reaction is 40 min, for first order reaction t1/2 is proportional to, the relation between t1/2 and k for first order reaction is,




0 件のコメント:
コメントを投稿